In 2013 I wrote an editorial for JBMT which I edit on the topic of the ‘dosage’ of applied manual therapies – entitled – “CAN WE DESCRIBE WHAT WE DO”
On September 28 2014 I posted the following on my facebook page:
“This is a gem from yesterday’s presentations in Rome (BioMech’O) :
Paul Standley PhD (University of Arizona, College of Medicine) discussed in vitro modelling of myofascial release (MFR) involving bioengineered tendons (BETs)- with and without fibroblasts. These were ‘wounded’ and the healing process was observed in relation to different versions of MFR using either 3% 6% 9% or 12% of stretch/elongation of the tissues, for 1,2,3,4,or 5 minutes.
- The optimal result in terms of wound closure was achieved (in comparison with other versions of MFR and with “no treatment” controls) with 6% of load for 3 minutes.
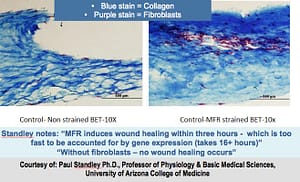
KEY POINTS:- Fibroblast-free BETs don’t heal
- Healing can be seen to start within 3 hours of MFR – too rapid for fibroblast gene-expression changes
- Fibroblast migration + collagen reorganisation seems to be the mechanism
- MFR, in time- and magnitude-dependent manners, differentially affect wound closure and wound shape change.
- !!!! Lowest magnitude MFR is without effect and highest magnitude is detrimental to wound healing.!!!!
- MFR-induced wound healing is associated with elevated fibroblast as well as collagen density at the wound site.
- This suggests that MFR initiates and/or accelerates cellular migration, cellular proliferation and collagen expression/secretion.
- Brief (1 minute), light (3%) MFR has no effect…strong (12%) long (5 minutes) aggravates the wound.
The photograph above shows a bioengineered tendon anchored at each end by protein scaffolding
The photograph below shows ‘artificial’ wound in artificial tendon, healing without (left) and with (right) load applied…..fibroblasts do their work better when appropriate degrees of tension/load is added.
The takeaway message therefore is that very small differences in degrees and timing of applied load (stretch in this example) make a huge difference in terms of how tissues respond via mechanotransduction.
Please note a key aspect of this study by Standley et al:
Dosed Myofascial Release in Three-Dimensional Bioengineered Tendons: Effects on Human Fibroblast Hyperplasia, Hypertrophy, and Cytokine Secretion
—– is that the healing response took place in the complete absence of any neurological features….the human fibroblasts responsible for starting the healing of the artificial/bioengineered (bovine collagen) tendon responded to tension/load, and started to close the wound.
https://leonchaitow.com/bookshop-europe/
https://leonchaitow.com/bookshop-usa/
Myofascial release…..Muscle energy techniques…..Positional release techniques (including strain/counterstrain)….and numerous other manual approaches achieve their effects via neurological AND mechanotransduction (and probably other) mechanisms…and therapists need to know about and understand that ‘dosage’ how much load, for how long … etc etc…is a key to successful intervention
- “Strain direction and duration of therapeutic load may influence pain modulating fibroblasts function, as well as inflammation and ranges of motion” (Standley & Meltzer 2008)
- Mechanical loading stimulates cellular synthesis promoting tissue repair and remodelling (Kahn & Scott 2009)
- Physical load (for example shear or compression) transduces into chemical signals within and among cells – altering metabolism, intracellular biochemistry and gene expression (Wipff & Hinz 2008)
- Collagen architecture responds to mechanical loading suggesting that therapeutic load may stimulate connective tissue repair an remodeling (Kjaer et al 2009)
REFERENCES
- Kjaer M 2009 From mechanical loading to collagen synthesis. Scand J Med Sci Sports 19(4):500-510
- Khan K Scott A 2009 Mechanotherapy. British J Sports Medicine 43:247–251
- Standley P Meltzer K 2008. Effects of Repetitive Motion Strain (RMS) & Counter-Strain (CS), on fibroblast morphology and actin stress fiber architecture. J Bodyw Mov Ther 12(3):201-203
- Wipff P Hinz B2008. Integrins and the activation of latent transforming growth factor beta1 – an intimate relationship. Eur J Cell Biol. Sep; 87(8-9):601-15
For more on these topics see:
https://leonchaitow.com/…/the-role-of-fascia-in-manipulativ…/
AND/OR
https://leonchaitow.com/…/…/the-explosion-of-fascia-research/
AND/OR
https://leonchaitow.com/…/21/fascia-congress-2009-early-not…/
AND/OR
https://leonchaitow.com/…/fascia-perspectives-clinical-mode…/ “